Welcome to Hope International University's IRB site, where you can find the resources to assist in research at and with our institution.
Hope International University (HIU) has a moral and legal responsibility to safeguard the rights, welfare, and dignity of human subjects involved in research. HIU is committed to the ethical principles for the protection of human subjects and is committed to ensuring that all human subject research, regardless of funding source. HIU's IRB policies comply with the Code of Federal Regulations (Title 45 Part 46 of the Code of Federal Regulations [45 CFR 46]) issued by the U.S. Department of Health and Human Services Office for Human Research Protections (OHRP) requirements as set forth in faculty, students, and staff who conduct research under the auspices of Hope International University must receive written IRB approval prior to initiating.
A vital safeguard of the privilege of conducting research involving human subjects is the institutional review of all research projects to minimize the possibility of unacceptable or unnecessary levels of risk to the rights, welfare, and dignity of human subjects. Careful review of this type also enhances the likelihood that any given research project will yield results that are accepted as valid by the scholarly community. Toward this end, and to comply with the requirements of federal law, HIU has created an Institutional Review Board (IRB). To assist the individual researcher in protecting the rights of human subjects and to minimize the potential legal liability of the investigator and the university should a human being be placed at risk, the IRB is instructed to review all projects involving human subjects.
Our IRB Policies, Procedures, and Resources are outlined in our IRB Handbook.
Review the IRB Handbook before completing an application to ensure you need to complete the IRB process and gain the information needed to help with the application.
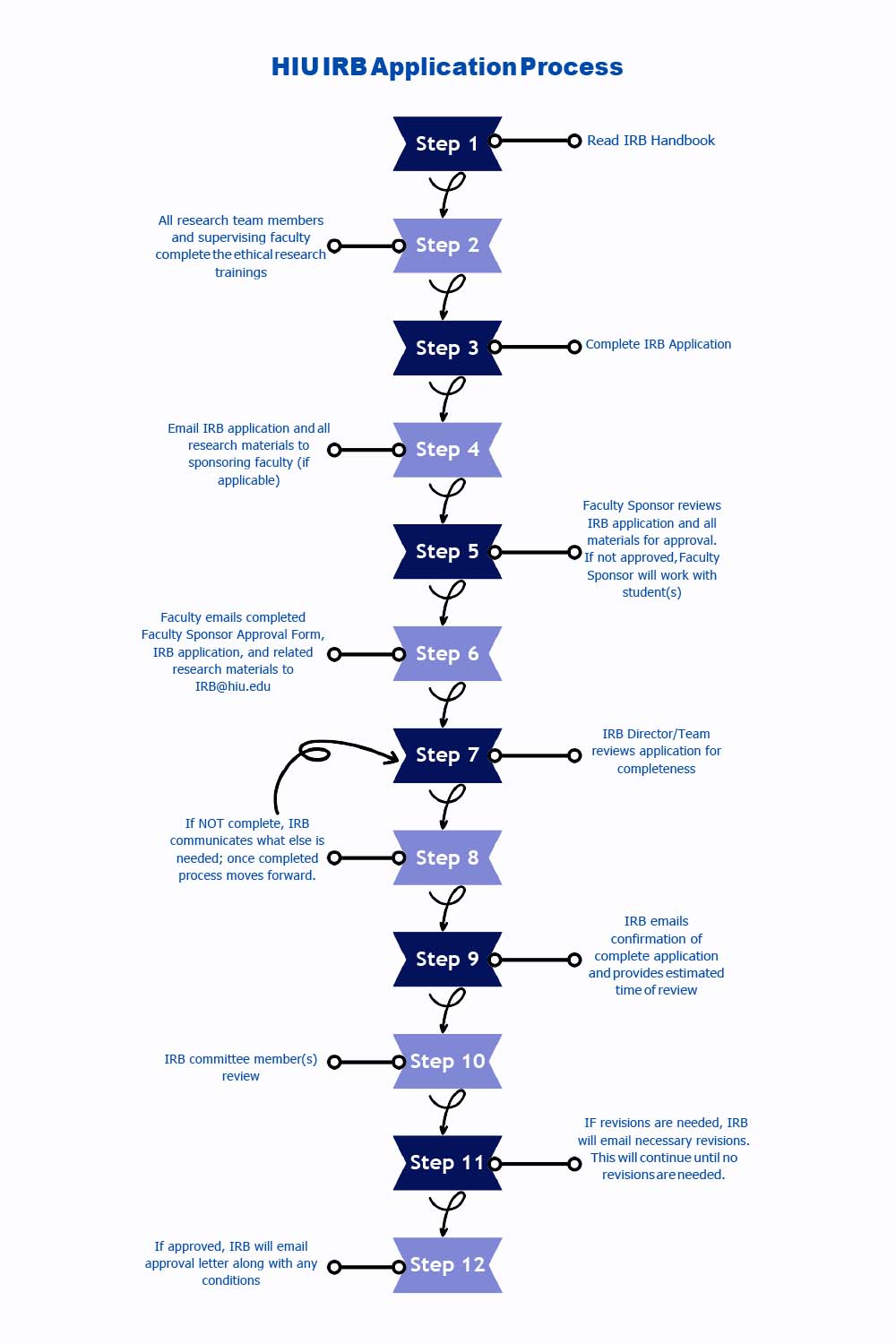
All Researchers: complete the "Social-Behavioral-Educational (SBE) Comprehensive" ethical human research training from the Collaborative Institutional Training Initiative (CITI). To get started, follow these steps:
Guide for CITI Registration: // Download //
Guide for Navigating the Training Course: // Download //

Make sure to download the application and use Adobe Acrobat Reader to complete IRB application.
Dissertation projects: your dissertation chair is your faculty sponsor.
Thesis projects: your thesis advisor is your faculty sponsor.
Research course projects: your instructor is your faculty sponsor.

Make sure to download the application and use Adobe Acrobat Reader or Pro to complete the Faculty Sponsor Approval Form.
Informed Consent: // Download //
Addendum for Children and Conservatees: // Download //